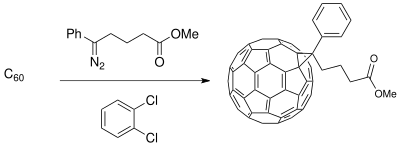
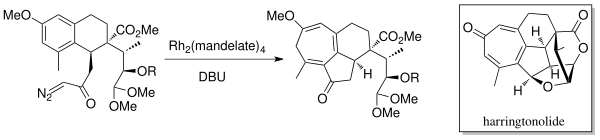
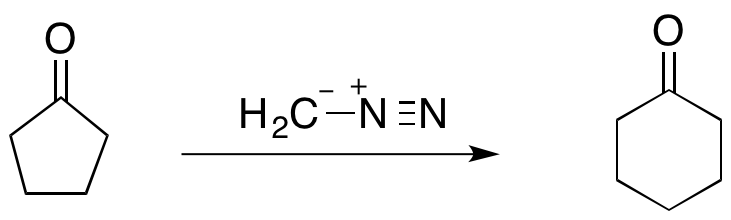Asymmetric Synthesis of a 5,7-Fused Ring System Enabled by an Intramolecular Buchner Reaction with Chiral Rhodium Catalyst | Organic Letters

Photochemically Switching Diamidocarbene Spin States Leads to Reversible Büchner Ring Expansions | Journal of the American Chemical Society
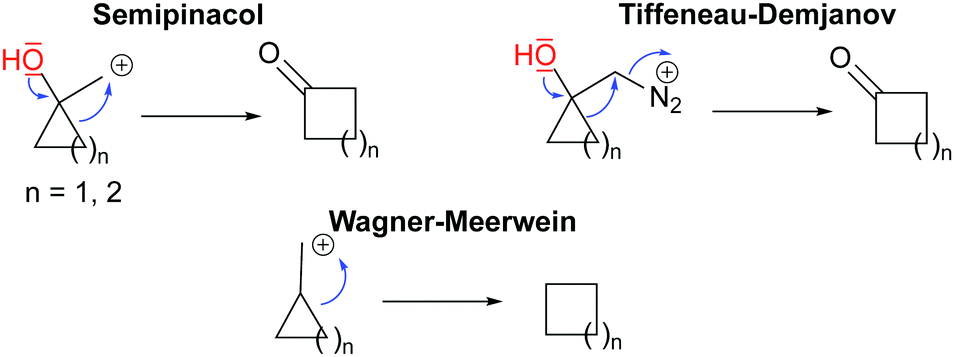
Small rings in the bigger picture: ring expansion of three- and four-membered rings to access larger all-carbon cyclic systems - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS01396J

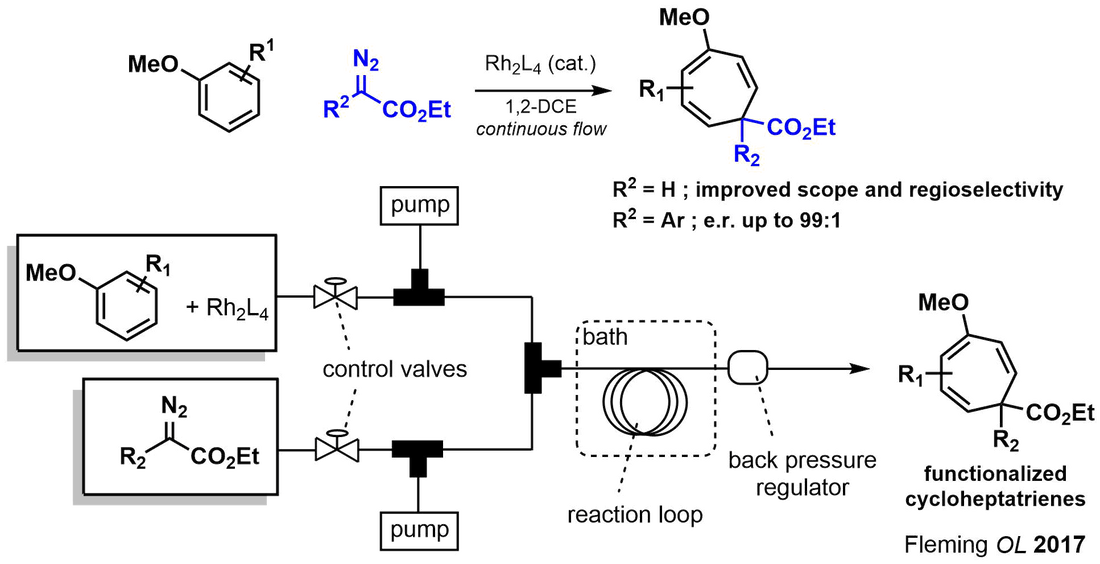
Regioselective and Enantioselective Intermolecular Buchner Ring Expansions in Flow | Organic Letters

Aza-Yang Cyclization—Buchner Aromatic Ring Expansion: Collective Synthesis of Cycloheptatriene-containing Azetidine Lactones | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Regioselective and Enantioselective Intermolecular Buchner Ring Expansions in Flow | Organic Letters
![Copper-catalyzed alkyne oxidation/Büchner-type ring-expansion to access benzo[6,7]azepino[2,3-b]quinolines and pyridine-based diones | Communications Chemistry Copper-catalyzed alkyne oxidation/Büchner-type ring-expansion to access benzo[6,7]azepino[2,3-b]quinolines and pyridine-based diones | Communications Chemistry](https://media.springernature.com/full/springer-static/image/art%3A10.1038%2Fs42004-023-00840-6/MediaObjects/42004_2023_840_Fig1_HTML.png)
Copper-catalyzed alkyne oxidation/Büchner-type ring-expansion to access benzo[6,7]azepino[2,3-b]quinolines and pyridine-based diones | Communications Chemistry

Aza-Yang Cyclization—Buchner Aromatic Ring Expansion: Collective Synthesis of Cycloheptatriene-containing Azetidine Lactones | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Regioselective and Enantioselective Intermolecular Buchner Ring Expansions in Flow | Organic Letters

Top: results for the Buchner ring expansion reaction between toluene 4... | Download Scientific Diagram

Hypervalent iodine promoted the synthesis of cycloheptatrienes and cyclopropanes - Chemical Science (RSC Publishing) DOI:10.1039/D1SC05429E

Photochemically Switching Diamidocarbene Spin States Leads to Reversible Büchner Ring Expansions | Journal of the American Chemical Society